
In order to identify the molecular mass of a compound, one can use its molar mass to identify the number of moles is in a sample of that compound. The Relevance Of Molar Mass Of O2 And Other Particles Water, for example, has a molecular weight of 18.015 atomic mass units, and one mole evaluates around 18.015 grams. One mole of H2O = 2 × 6.02214076 × 1023 of Hydrogen + 6.02214076 × 1023 of Oxygen.īriefly summarized, the mass that comprises one mole of a substance will amount to its molecular weight. Right here is an instance of how moles can streamline chemical representation and also calculation. In order to comprehend precisely how chemicals interact and how molecules within chemical examples interact, Avogadro’s number should made use of. 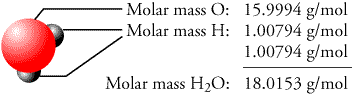
It completes so that weight can be compared with the variety of atoms within the material (as specified by the number of moles) since weight is much easier to track than the number of atoms in the chemical sample.Ĭomputations including more than one chemical call for the use of Avogadro’s number as well as moles. The chemical reactions include billions of atoms engaging with one another and being reorganized nevertheless, it would be impossible to represent the motion of billions of atoms.ĭespite that, scientists still require a unit of dimension that can stand for billions of elementary entities. Therefore, the number of atoms is 6.02214076 × 1023, which is called Avogadro’s number.Īvogadro’s number and the mole are essential principles in chemistry. There coincide several elementary entities within one mole of carbon 12 as there are atoms.  
Atoms and also molecules are basic entities. Scientists utilize moles to determine the variety of elementary entities found in an example of chemicals. A mole is a device of dimension that is utilized to specify the amount of material. When it comes to moles, however, this meaning does not help a lot. The molar mass of any chemical compound is the mass that one mole of that substance possesses, the group that one mole of an offered material keeps. The Relevance Of Molar Mass Of O2 And Other ParticlesĪn aspect’s Molar Mass Of O2 is defined as the amount of the masses of its essential components.If we discuss a mole of an ionic substance, we will still use molar mass. Ionic substances do not have private molecules. When describing substances that are not molecular (ionic compounds), the term “molecular mass” is improper, and also “formula mass” is generally utilized instead. Put merely, N2 has a molar mass of 28.02 grams per mole. This outcome is called the molecular mass of a molecule. Let’s learn more about Molar Mass Of O2.Īn atomic mass is simply the sum of the atomic masses of all its component atoms. As for nitrogen, its atomic mass is merely (14.01 + 14.01) = 28.02 amu. For a particle (for instance, nitrogen, N2), the mass amounts to the sum of the atomic masses of both nitrogen atoms. Compounds can likewise be determined utilizing the idea of molar mass. Knowing what molar mass is and exactly how it belongs to doing computations in chemistry will help us recognize why understanding Oxygen’s molar mass is important. It is essential to recognize Oxygen’s molar mass since eight is the atomic number of Oxygen, and the molar mass of O2 is approximately 15.9994. Molar Mass Of O2 (Oxygen Particle): In deep space and on Earth, Oxygen is one of the most abundant aspects.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
